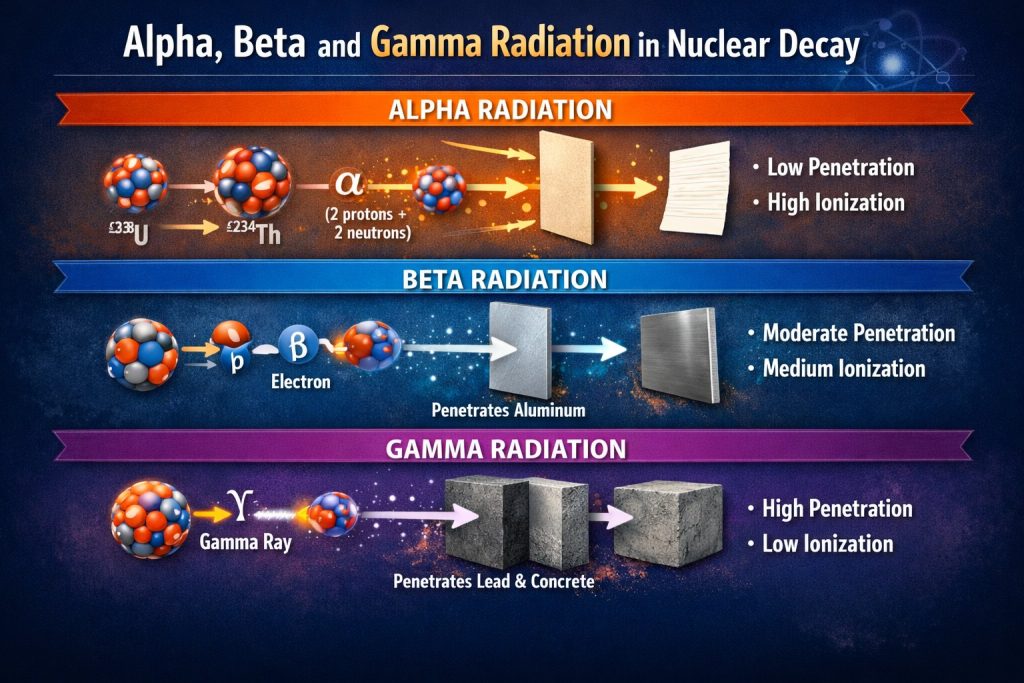
When an unstable atomic nucleus transforms itself into a more stable configuration, the process is known as nuclear decay. During this transformation, energy is released in the form of radiation. The three most common types observed in natural and artificial radioactive processes are alpha radiation, beta radiation, and gamma radiation. Although these forms of radiation often appear together, they are fundamentally different in nature, behavior, and impact on matter. Understanding these differences is essential in physics, medicine, nuclear engineering, and radiation protection.
Alpha Radiation
Alpha radiation consists of alpha particles, which are composed of two protons and two neutrons. In essence, an alpha particle is identical to the nucleus of a helium atom.
During alpha decay, a heavy and unstable nucleus ejects this particle to reduce internal instability. As a result, the original atom transforms into a different element with a lower atomic number.
Key characteristics of alpha radiation
• High mass and positive charge
• Very low penetration ability
• Strong ionizing effect over short distances
Alpha particles are easily stopped by a sheet of paper or even the outer layer of human skin. However, if alpha emitting material is inhaled or ingested, it becomes extremely dangerous due to intense local ionization within living tissue.
Alpha decay typically occurs in heavy elements such as uranium and radium.
Beta Radiation
Beta radiation is produced when a nucleus contains an imbalance between protons and neutrons. To correct this, a neutron can transform into a proton, or a proton into a neutron, releasing a beta particle in the process.
There are two forms of beta decay
• Beta minus decay releases an electron
• Beta plus decay releases a positron
Beta particles are much smaller and lighter than alpha particles and carry a single electrical charge.
Key characteristics of beta radiation
• Moderate penetration capability
• Medium ionizing strength
• Can penetrate skin but is stopped by thin metal layers
Beta radiation is commonly encountered in medical imaging, industrial thickness measurements, and laboratory research. Proper shielding is required to prevent skin exposure and secondary radiation effects.
Gamma Radiation
Gamma radiation is fundamentally different from alpha and beta radiation. It does not consist of particles but of high energy electromagnetic waves. Gamma rays are emitted when an excited nucleus releases excess energy after undergoing alpha or beta decay. The atomic structure remains unchanged, but the energy state decreases.
Key characteristics of gamma radiation
• No mass and no electric charge
• Extremely high penetration power
• Lower ionization per interaction but deep tissue reach
Gamma radiation can pass through the human body and requires dense materials such as lead or thick concrete for effective shielding. Because of its penetration depth, gamma radiation is widely used in medical diagnostics, cancer therapy, and industrial inspection.
Comparison of Radiation Types
Alpha radiation interacts strongly with matter but travels only short distances.
Beta radiation travels further and requires light shielding.
Gamma radiation penetrates deeply and demands heavy shielding.
Each type plays a different role in science and engineering, and each carries distinct risks depending on exposure conditions.
In which fields is nuclear decay relevant?
It underpins technologies such as
• Medical imaging and radiotherapy
• Nuclear power generation
• Archaeological dating methods
• Radiation safety engineering
A precise understanding of alpha, beta, and gamma radiation allows engineers and scientists to harness nuclear processes responsibly while minimizing risk to humans and the environment.